  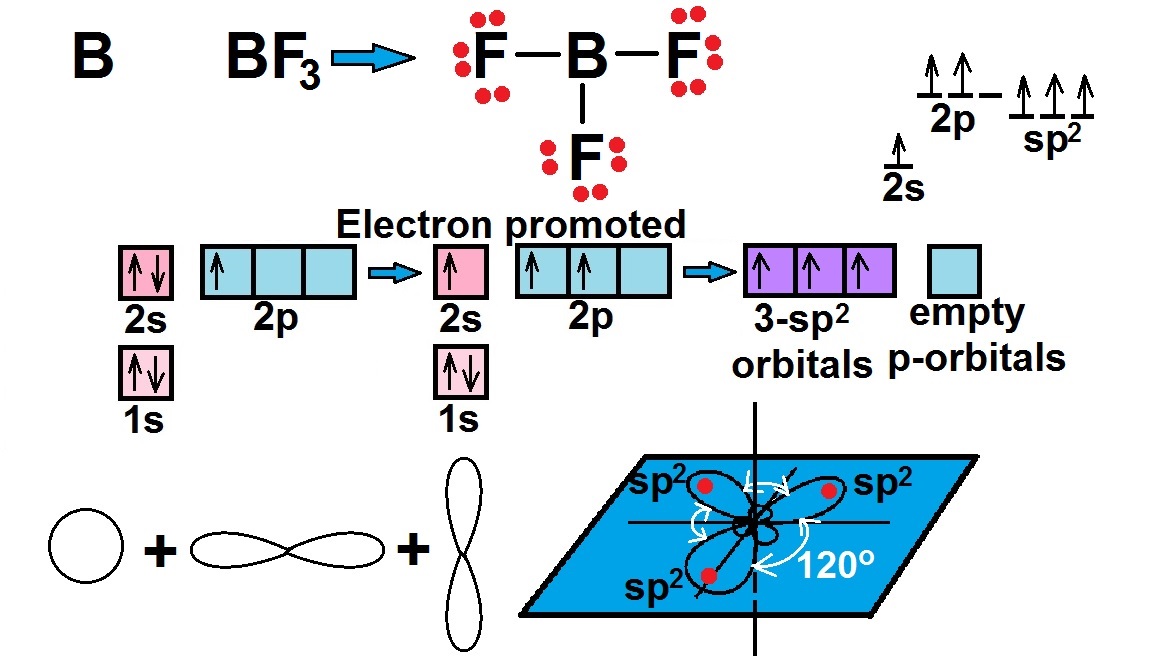
Fluorine too completes its last shell by making a bond with boron’s electrons.Īs per the molecular configuration of BF3, atoms of B take p-orbitals and s-orbitals turning into sp2 hybrid orbitals through a special process called hybridization. Thus, as per our collected data above, each fluorine must have 8 electrons to complete its octet and boron must have 6 electrons in total for marinating their stability.Ĭonsidering the exceptional case of boron, fluorine shares its electrons with boron to have 6 electrons. Hence, B makes 3 single bonds (6 electrons) with atoms of F.Īssign the remaining 18 valence electrons around in a way that each F consists of 6 electrons surrounding it. To get the number of lone pairs, calculate the difference between the number of valence electrons and the number of bonding pairs electrons.īefore placing electrons around the atoms to complete the octets, select the central atom.Īs per arrangements of elements in the table, B is the least electronegative than F. Now, find the total number of bonding electrons that are used for bonding in the Lewis dot structure.Ĭompute the difference between required electrons and total valence electrons.Ħ number of bonding pairs of electrons is equal to 3 covalent bonds formed between F and B atoms. To compute the required number of electrons that must share by each atom, F needs 8 electrons in its outermost shell and B needs most 6 valence electrons in its last shell. After computation, the result says that 24 valence electrons need to be shared to make a bond between atoms. In the compound, there is 1 atom of boron and 3 atoms of fluorine. It needs 6 valence electrons in its outermost shell. Hence, you can calculate the number of valence electrons of both the atoms using electronic configuration.Īs per the octet rule, born is an exceptional case. The atomic number of boron (B) and fluorine (F) is 5 and 9 respectively.Lone pairs (non-bonding pairs: when electrons do not participate in the formation of bonding between atoms).Required number of electrons to complete octet.To learn about any Lewis dot structure of boron trifluoride BF3, you need to compute mainly four important things. The periodic table helps you to study various elements that include atomic number, valency, etc. Sharing electrons on each atom make a covalent bond.Make sure you follow the octet rule that says each atom must complete its last shell consisting of a total of 8 electrons.At first, make a single bond with the central atom.Identify the least electronegative atom and put it in central space.Find out the lone pairs (number of unpaired electrons) and bonding electrons (electrons that take part in the formation of a bond).Sum the product of the number of valence electrons of an atom to the number of the same atoms present in the molecule to get the total number of valence electrons of the compound.Count and calculate the number of valence electrons present in the last shell of an atom.Study the features of the periodic table that helps in finding the atomic number and informs about the electronegativity of the elements.Conclusion Points Related to Lewis Structure Other elements that may at times form incomplete octets include Beryllium and Aluminum.īF3 Ball and Stick Model. BF3 for example will try to form an octet on the central atom by converting to BF4. However, when these kinds of elements are handled in real life they are often unstable due to the incomplete octet. Therefore, it is simply assumed that the structure with the incomplete octet is a more stable configuration. Since double-bonding one of the fluorine's to complete the octet would leave the fluorine with a formal charge of +1, it becomes quickly apparent the infeasibility of this structure since fluorine is the most electronegative element and would not simply lose an electron. These are generally called incomplete octets and are formed due to the low electronegativity of Boron. You may have observed that this structure for BF3 seems to violate the octate rule specifically for the central Boron atom which only has six electrons in its valence shell.  
This results in an overall structure that is generally nonpolar in character. Answer: BF3 is nonpolar because all of the fluorine atoms are arranged in a trigonal planar fashion around the molecule, thereby cancelling out each other's pull for electrons.Īlthough the difference between Boron's electronegativity (2.04) and Fluorine's (3.98) is great enough to make the individual bonds polar covalent, the symmetrical arrangement of atoms around the central Boron atom cancels out these possible dipole moments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
